IgG is an abbreviation for immunoglobulin G. IgG is in the antibody family of proteins along with IgM, IgA, IgD, and IgE. Antibodies are proteins made by immune cells to recognize foreign objects in the body. They are secreted into the blood and circulated throughout the body so they can find those foreign objects. Each IgG is specially made to hunt and bind to a specific foreign object, called its antigen. For example, an anti-Spike IgG specifically binds to its antigen, Spike.
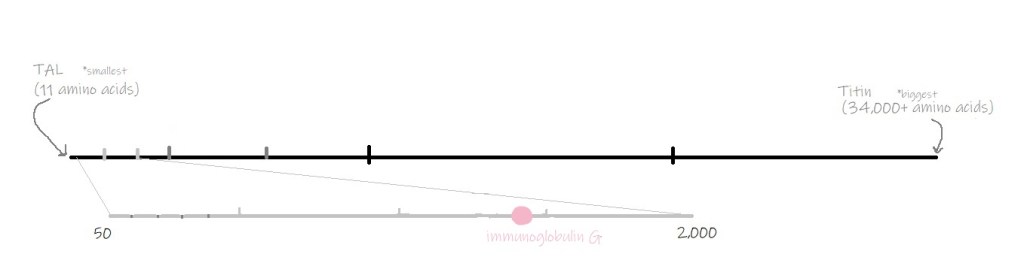
IgG is made up of two heavy pieces (each 440-550 amino acids) and two light pieces (each 220 amino acids). Each of these four pieces has what is called a variable region, which is different on every IgG. The variable region is the part of the IgG that binds to its antigen. Other proteins in the antibody family have similar structures, and IgM and IgA are made up of several “copies” of this structure.

Immune cells design new antibodies by mixing-and-matching different versions of IgG genes. Like all proteins, IgG is made using instructions in your DNA. Any particular IgG recipe is made from four sets of instructions, named for its different parts: V, D, J, and C. The C segment is constant; it is used in all IgG recipes. Your DNA contains many options for V, D, and J instructions. When immune cells design a new antibody, they choose one V, one D, and one J segment to build the variable region, then use the constant C segment for the rest. There are millions of possible recipes that your immune cells could put together to make all kinds of different IgG’s.

Antibodies like IgG are incredibly useful in scientific research because (1) they can be made to bind any antigen desired and (2) they only bind to their antigen, and nothing else. They can be made by animals like mice or rabbits, and now they can be made in cell cultures also. Once they are gathered and purified, IgG’s are used everyday by scientists around the world to detect specific antigens in a variety of situations.
For example, in my lab, I use IgG’s to look at muscle proteins under the microscope. First I attach a fluorescent dye molecule to IgG’s, and then I allow them to bind to their target protein in muscle fibers. When I put those muscle fibers under the microscope, the light emitted by the attached fluorescent dye shows me where in the muscle fiber my protein of interest is located.
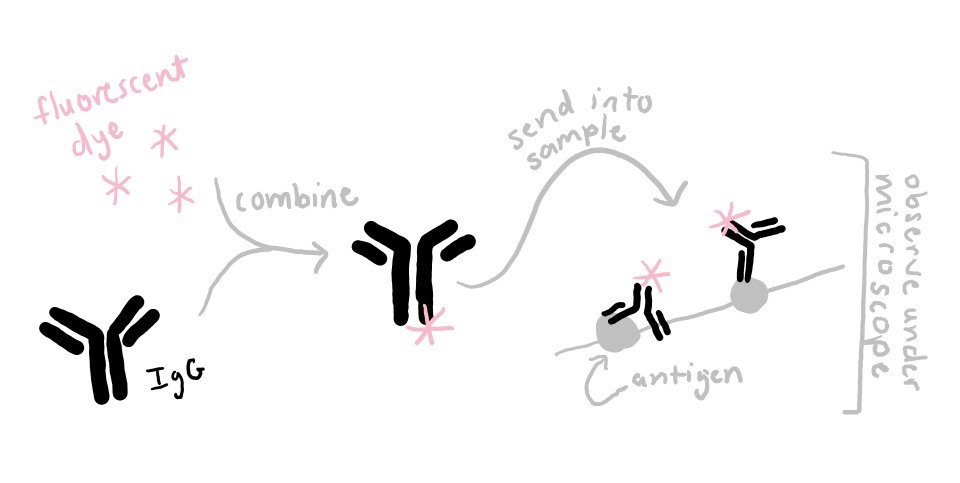
IgG’s can also be used to detect the presence of a molecule, to purify a particular protein, to measure the molecular weight of a protein (look up: Western Blot), to discover protein binding partners, or to determine the location of a molecule. You can even use IgG’s at home to test your urine for signs of drug use or pregnancy!
Extra reading:

