It often goes by the nickname “APP” for short. APP has been extensively studied because of its association with Alzheimer’s disease. Despite being studied so intensely, APP’s role is complex and there is a lot that researchers just have not figured out yet. (But I will share some of what we do know!)
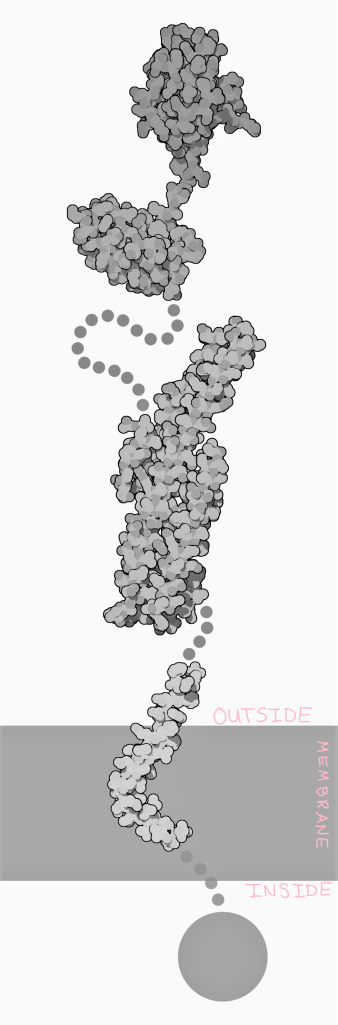
APP lives in the membranes of brain cells. We have not been able to see what exactly it looks like as a whole, but we have been able to see some of its parts. Like many membrane proteins, APP has a region outside of the cell, a region inside of the cell, and a region inside of the membrane.
The function of APP is also not well understood yet, but it is clear that APP is involved in several different operations. APP is possibly a receptor–a protein that relays a signal from outside the cell to inside the cell. APP is also possibly involved in sticking cells (specifically neurons) together. The outside of an APP from one cell can stick to the outside of an APP from another cell.
APP is not only important for its own jobs, though. It is cleaved into several pieces, each of which have their own jobs, too. Some pieces control the growth of neurons, formation of synapses (connections between neurons), building certain proteins in the cell, and loading cargo for transport across the cell. We can thank APP and its pieces for the ability to think and form new connections in our brains!
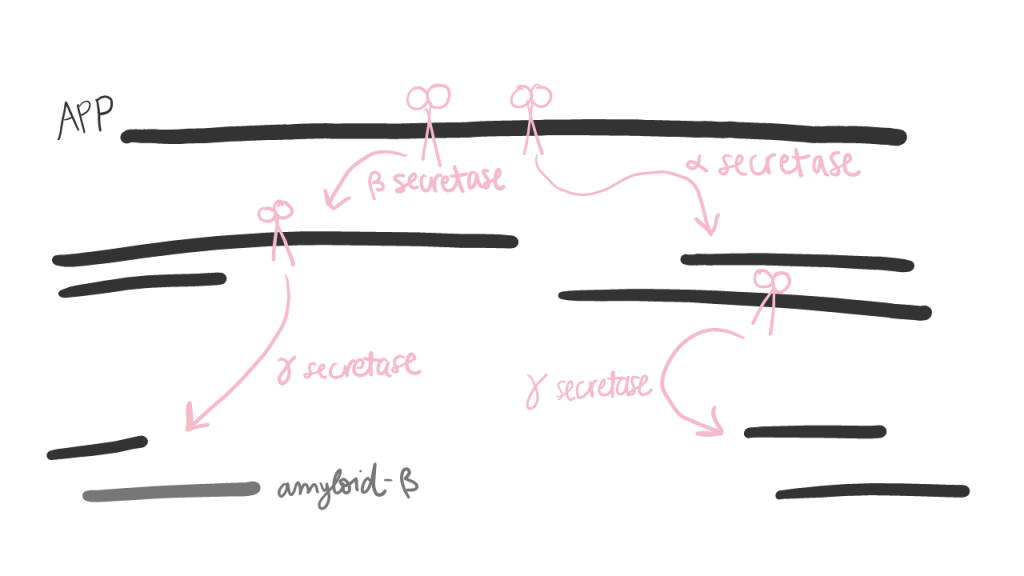
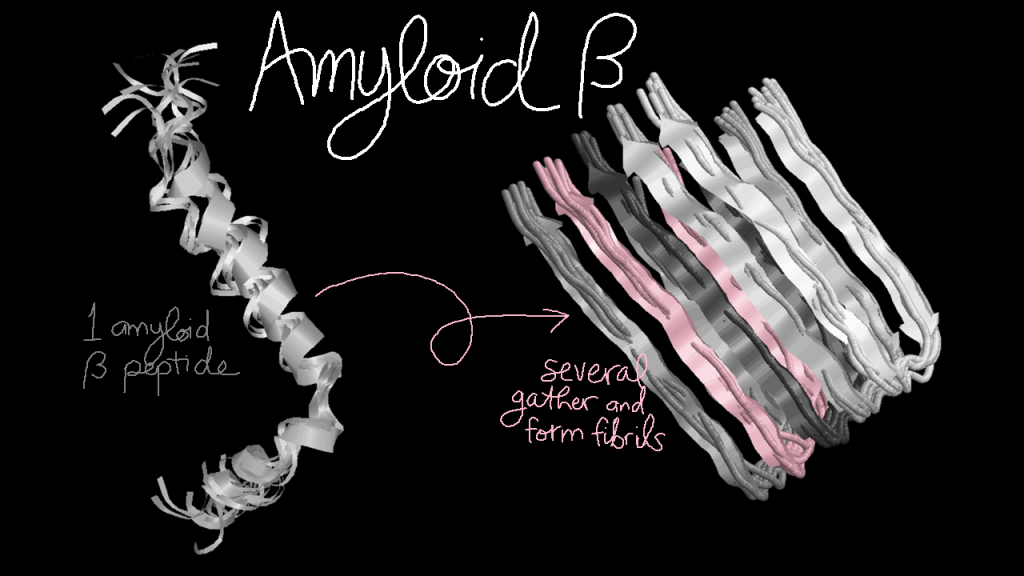
One of the pieces that results from APP cleavage is the infamous amyloid-beta peptide. Most amyloid-beta peptides operate normally, but mutations in this peptide can make them extra sticky. Many mutated amyloid-beta peptides will stick together and form fibrils, which can grow into larger plaques. These plaques are associated with Alzheimer’s disease, but it is not 100% clear whether they are a cause of the disease or a symptom… or both.
One of APP’s many potential jobs is controlling amounts of amyloid-beta peptide. Amyloid-beta peptide can bind to APP and cause a change, so when amyloid-beta peptide builds up too much, APP sends a signal to slow down amyloid-beta peptide production.

One might predict that in Alzheimer’s patients, APP is abundant. After all, the amyloid fibrils have to come from somewhere, right? But interestingly enough, researchers have actually found that Alzheimer’s patients tend to have lower amounts of APP. It is possible that lower levels of APP, which is involved in forming new connections in the brain, contributes to dementia.
There is a lot left to research, but one thing is for sure: it is critical that we deepen our understanding of APP and its functional pieces. How is the amount of APP in your cells controlled? Does that control change as people age? How? What exactly does APP do and what other proteins does it interact with to do those things? And perhaps most important of all, how can we prevent mutant amyloid-beta peptides from damaging the brains of our loved ones? If you are interested in science and you are looking for a field of research, there is so much to do here. If you aren’t looking to get into research, you can still help by donating to Alzheimer’s research.
Zheng, H., Koo, E.H. Biology and pathophysiology of the amyloid precursor protein. Mol Neurodegeneration 6, 27 (2011). https://doi.org/10.1186/1750-1326-6-27

