Meet Myosin Binding Protein C!
Myosin Binding Protein C’s childhood nickname was “C protein,” but these days it goes by “MyBP-C.”
MyBP-C is a regulatory protein in muscle. As its name implies, it binds to myosin, which is the protein responsible for producing the force in a muscle contraction (meet myosin here!). It also binds to actin, the cytoskeletal protein that myosin pulls on (meet actin here!). MyBP-C can bind to myosin and actin in different ways, and each of those ways have a different effect on muscle contraction.
MyBP-C has a structure like “beads on a string.” There are NUMBER of “beads” (plus an extra one in cardiac muscle). The beads are linked by skinny strings we call “linkers.” A whole MyBP-C looks something like this:
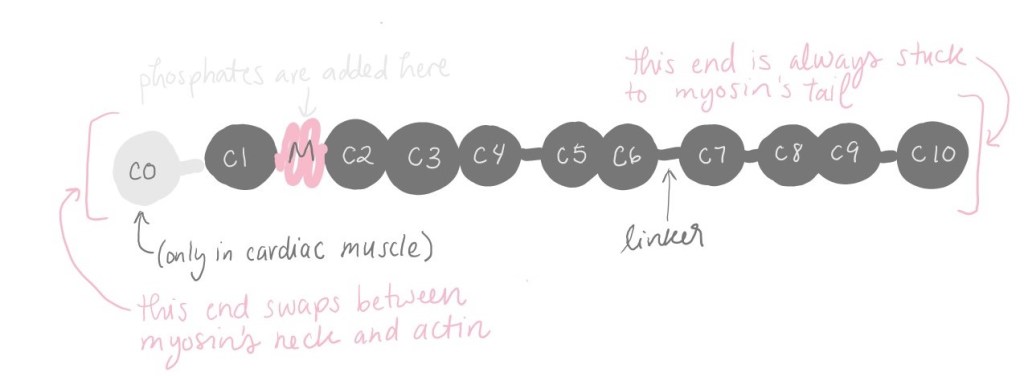
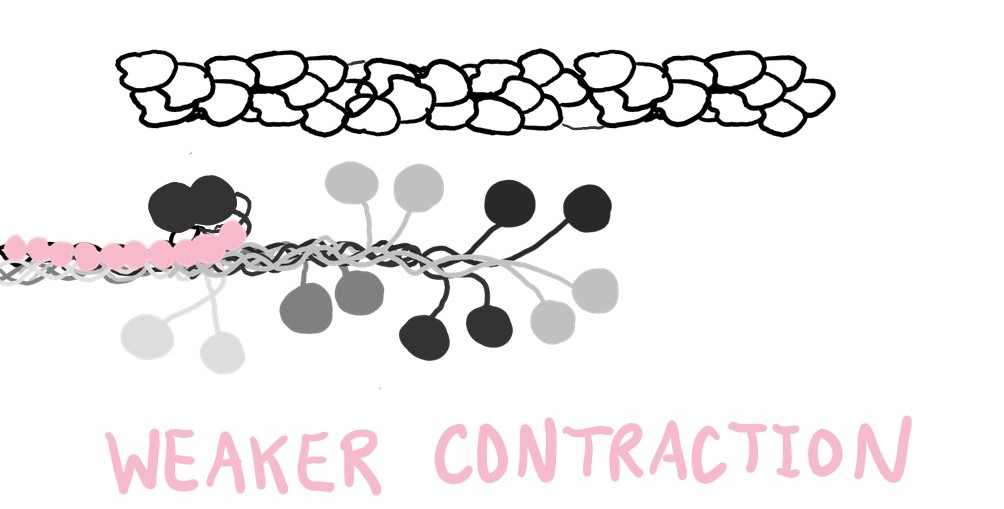
On one end, MyBP-C sticks to myosin’s tail, and it doesn’t let go. MyBP-C can use its other end to stick to either myosin’s neck or actin. When MyBP-C binds to a myosin’s neck, that myosin is encouraged to turn “off,” to take a break, to clock out. It doesn’t contribute to muscle contraction, so the contraction is weaker.
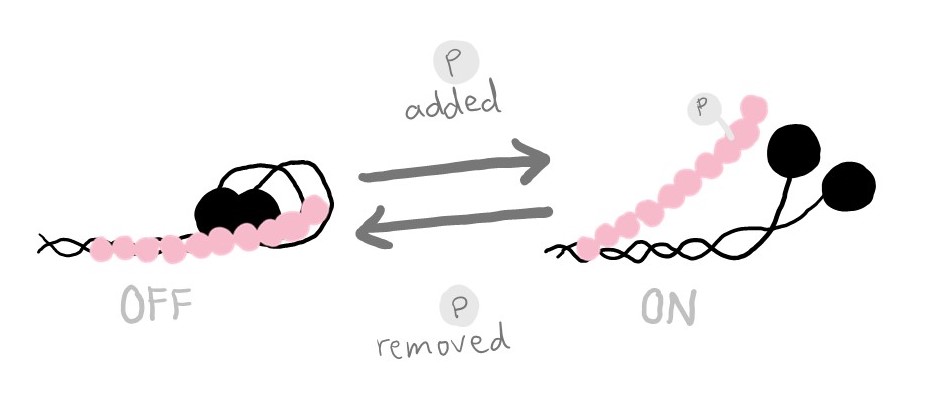
Signals to the muscle cell can trigger a change to MyBP-C that also changes its current job. As the signal is relayed throughout the cell, a negatively-charged molecule called a phosphate is added onto MyBP-C’s biggest linker. The new addition causes MyBP-C to readjust its structure a bit, and the adjusted MyBP-C no longer sticks to myosin’s neck. It would rather stick to actin.
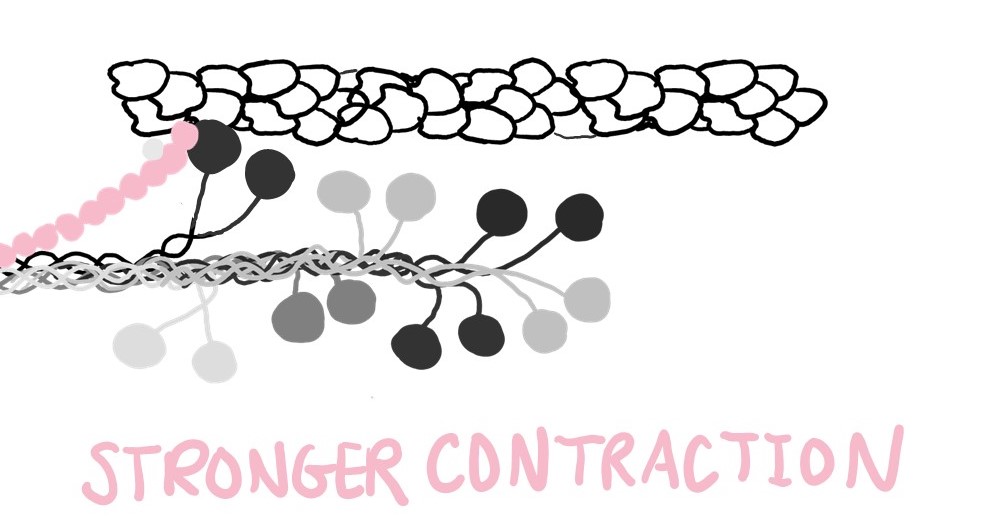
Now that myosin’s neck is released, it can return to work. Its contribution to muscle contraction makes the contraction stronger.
It may sound nice to have strong contractions but in reality you want a sweet middle spot, especially in your heart muscle. Mutations in MyBP-C are connected to hypertrophic cardiomyopathy, a heart disease that results from too much contraction. Just like working your arm and leg muscles will cause them to grow, this heart disease constantly overworks the heart muscle causing it to grow. As the heart walls get bigger, the room inside for blood gets smaller, and though the heart works hard to bump blood, it cannot pump enough at a time. Studying muscle proteins like MyBP-C can help scientists (like me and my lab mates!) develop treatments for heart diseases like this one!

