Meet Voltage-Dependent Anionic Channel! Scientists call it by its nickname, VDAC.
VDAC lives in the mitochondria (that’s right: the powerhouse of the cell), specifically in the outer membrane. It’s the most abundant protein in the outer mitochondrial membrane and it is present in every single mitochondrion on earth.
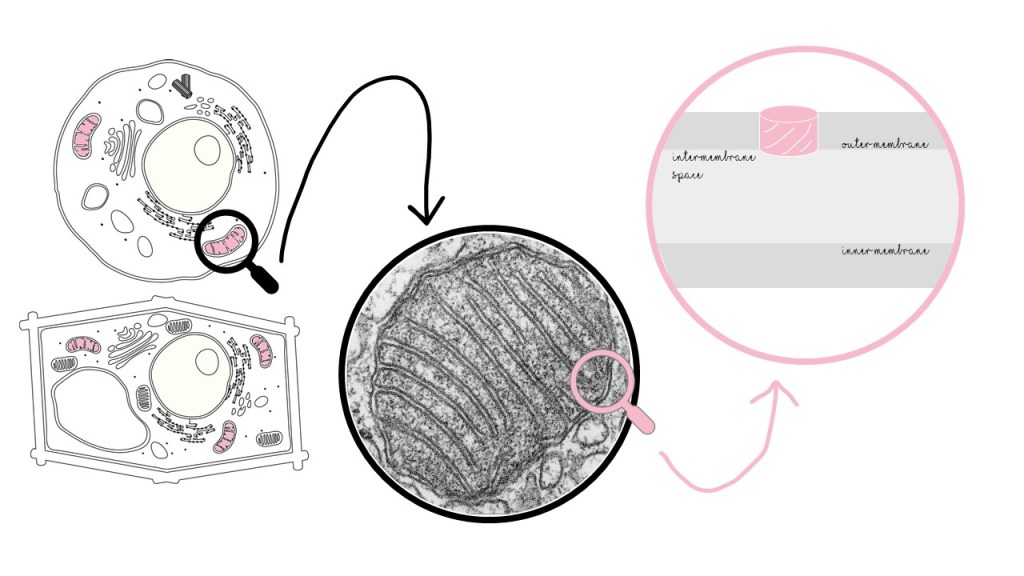
Mitochondria supply energy to the cell in the form of ATP. As a channel, VDAC has a critical role in mitochondria: it behaves like a gate, allowing ATP to exit and go to the rest of the cell. It also lets in ADP, a required material for ATP production. Without VDAC, the mitochondrial “powerhouse” would be a power plant with no powerlines and no fuel: useless!
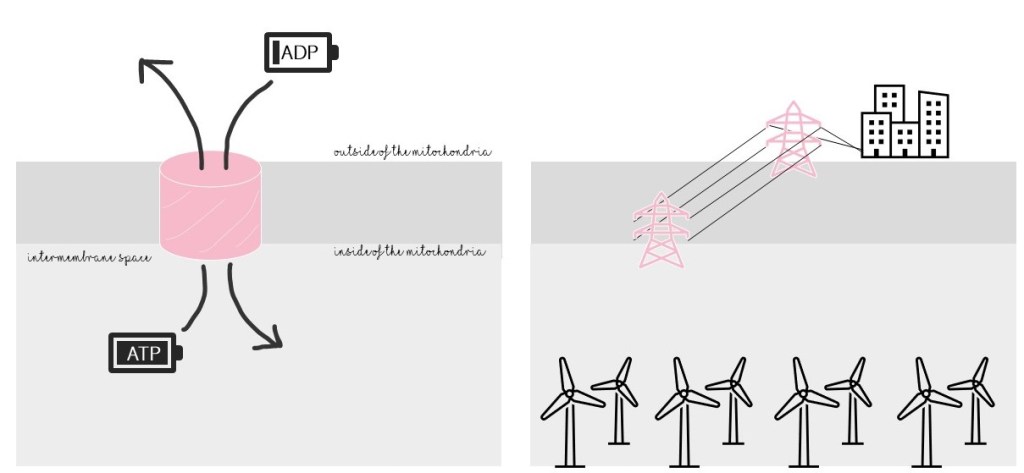
VDAC also allows other negatively-charged molecules (besides ATP and ADP, which are also negatively charged) to go in or out of the mitochondria, though these molecules are less critical and less studied. Another word for “negatively charged” is “anionic,” fitting VDAC’s name.
VDAC has a classic structure known as a beta-barrel, which is just a series of beta sheets (you can read about those here) arranged in a barrel shape! You may have seen a beta-barrel before: Green Fluorescent Protein. The inside of the barrel is positively-charged to attract the negatively-charged molecules that pass through it. The outside of the barrel is hydrophobic to allow it to sit in the hydrophobic membrane. VDAC also has a small helix… but its function is an ongoing debate.
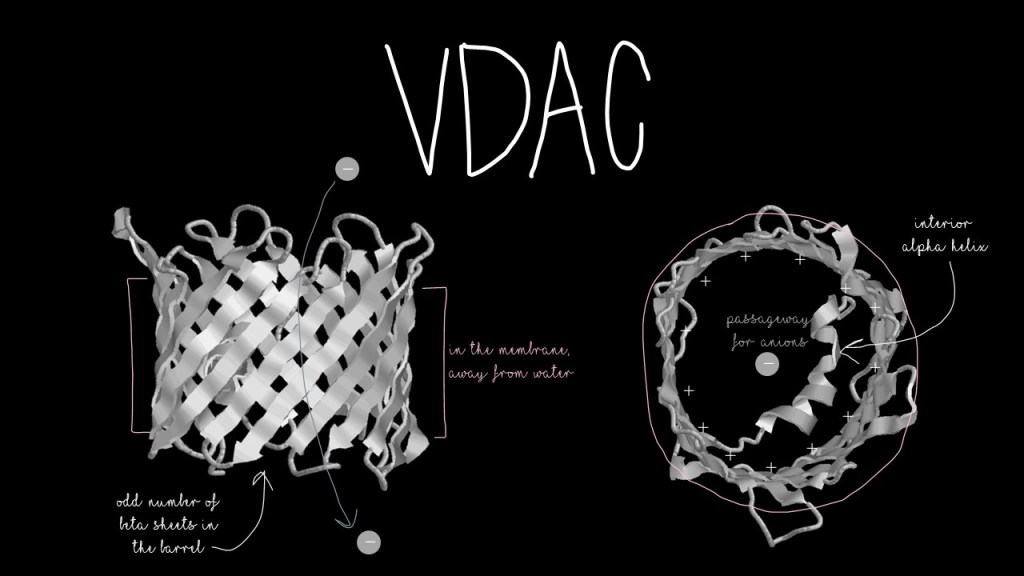
One would assume, based on its name, that VDAC is a channel for anions, negatively-charged molecules, that depends on voltage for opening and closing. Several channel proteins read changes in voltage to decide whether to open or close. It turns out, VDAC is not dependent on voltage at all. So why did people think that it was? To answer that, we have to look back to its structure.
Most beta barrels found in animals have even numbers of beta-sheets. Bacterial beta barrels tend to have odd numbers. Count up the sheets in VDAC, and you will see that it has nineteen–an odd number. VDAC is more closely related to bacterial outer membrane proteins than it is to other plant or animal beta barrels! And many of these related bacterial protein channels are voltage-dependent. Another VDAC relative, TOM40, lives in the same place, has similar origins, and is voltage-dependent. VDAC and TOM40’s relationship to bacterial outer membrane proteins points toward the theory that mitochondria originated from the engulfing of a bacterium by another cell.
Its misnomer is only the smallest of VDAC’s mysteries.
VDAC is involved in activating cell death by allowing a protein signal, cytochrome c, to flood out of the mitochondria. Inhibiting VDAC inhibits cytochrome c’s exit. Cytochrome c crosses man-made membranes with only VDAC. But cytochrome c can’t fit through VDAC’s opening. How then does VDAC help cytochrome c cross the membrane? Understanding VDAC’s involvement in activating cell death could help us develop treatments for cancer.
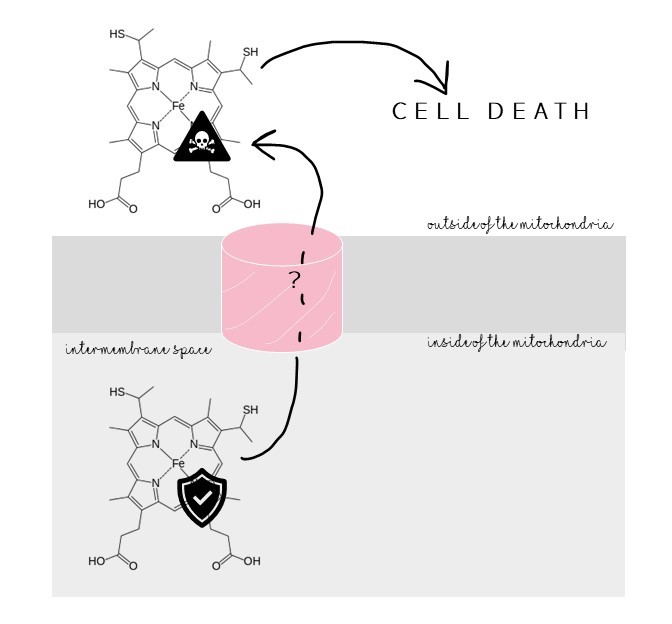
VDAC has been found in pairs, but why? And how do they stick together?
VDAC has a curious negative charge that sticks into the membrane. Negative charges don’t belong in the neutral, uncharged, hydrophobic environment of the membrane! What role does this charge have? How does it disrupt the membrane around it?
Remember that the role of VDAC’s helix is up for debate? And we still don’t even know what the channel looks like when it is closed!
There is obviously a lot to learn about VDAC and this field is incredibly active. Scientists like Yessica (@mintyestationery on Instagram) and her colleagues are making great strides to get to know VDAC so that we can better understand the powerhouse of the cell and a protein we literally cannot live without.
Thank you to Yessica for chatting with me and answering all of my questions about VDAC!
Koebnik, R., Locher, K. P., & Gelder, P. Van. (2000). Structure and function of bacterial outer membrane proteins: barrels in a nutshell. Molecular Microbiology, 37(2), 239–253. https://doi.org/10.1046/J.1365-2958.2000.01983.X
Madesh, M., & Hajnóczky, G. (2001). VDAC-dependent permeabilization of the outer mitochondrial membrane by superoxide induces rapid and massive cytochrome c release. Journal of Cell Biology, 155(6), 1003–1016. https://doi.org/10.1083/JCB.200105057
Poynor, M., Eckert, R., & Nussberger, S. (2008). Dynamics of the Preprotein Translocation Channel of the Outer Membrane of Mitochondria. Biophysical Journal, 95(3), 1511–1522. https://doi.org/10.1529/BIOPHYSJ.108.131003

